Phenoxy-Linked Colchicine Derivatives: A Structure-Based Approach toward Enhanced Selectivity and α-Tubulin Interaction
Link: https://doi.org/10.1016/j.ejmech.2026.118732
Title: Phenoxy-Linked Colchicine Derivatives: A Structure-Based Approach toward Enhanced Selectivity and α-Tubulin Interaction
Authors:
Gbenga Olorunmodimu, Darius Vrubliauskas, Jantana Yahuafai, Duangjai Todsaporn, Borwornlak Toopradab, Phornphimon Maitarad, Thanyada Rungrotmongkol, Chanat Aonbangkhen, Tanatorn Khotavivattana
Abstract:
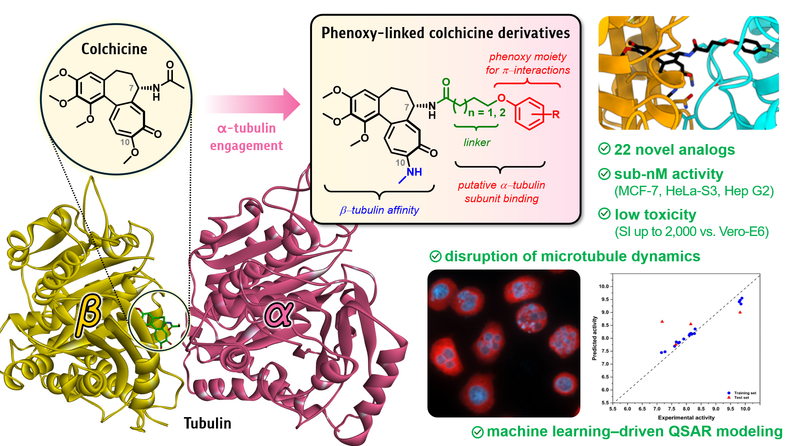
Colchicine, a well-known microtubule assembly inhibitor that binds to β-tubulin, has limited clinical utility due to high systemic toxicity. To enhance its therapeutic window, 22 novel phenoxy-linked colchicine derivatives were designed and synthesized, based on the hypothesis that additional engagement of α-tubulin could enhance both potency and selectivity. Antiproliferative evaluation across MCF-7, HeLa-S3, and Hep G2 cancer cell lines identified derivatives with remarkable IC50 values (as low as 0.08 nM in MCF-7), showing up to 7-fold and 13-fold greater potency compared to colchicine and doxorubicin, respectively. Selectivity was evaluated against Vero E6 and human MRC-5 normal cells, with several derivatives maintaining favorable selectivity indices; for instance, compound 6c achieved a selectivity index (SI) of 210 in MCF-7 cells (8-fold vs. colchicine) and 2000 in HeLa-S3 cells (77-fold). Molecular docking supported α-tubulin interactions, and immunofluorescence assays confirmed disruption of microtubule dynamics. Furthermore, machine learning–driven quantitative structure–activity relationship (QSAR-ML) modeling using gradient boosting regression (GBR) demonstrated strong predictive performance, while SHapley Additive exPlanations (SHAP) analysis revealed key structural features associated with anticancer activity and normal cell toxicity. These findings establish α-tubulin engagement as a promising strategy for the development of highly potent and selective tubulin inhibitors, offering new opportunities in cancer therapy.
Cite this:
Olorunmodimu, G.; Vrubliauskas, D.; Yahuafai, J.; Todsaporn, D.; Toopradab, B.; Maitarad, P.; Rungrotmongkol, T.; Aonbangkhen, C.; Khotavivattana, T. "Phenoxy-linked colchicine derivatives: A structure-based approach toward enhanced selectivity and α-tubulin interaction" Eur. J. Med. Chem. 2026, 309, 118732. https://doi.org/10.1016/j.ejmech.2026.118732




